SPRAVATO® (Esketamine) Treatment
Spravato® Treatment for Depression in League City, TX
FDA-Approved Esketamine Therapy for Treatment-Resistant Depression at Family Psychiatry
Family Psychiatry in League City, TX offers SPRAVATO® (esketamine) nasal spray, an FDA-approved treatment for adults with treatment-resistant depression (TRD) and for adults with major depressive disorder (MDD) experiencing acute suicidal ideation or behavior. SPRAVATO® is administered under close medical supervision at our certified treatment facility located at 2360 Gulf Freeway S, Suite 100B, League City, TX 77573. All Spravato treatments are evaluated and overseen by Dr. Mohammad Saeed, M.D., a triple board-certified psychiatrist with over 40 years of clinical experience in the Greater Houston area. We serve patients from League City, Clear Lake, Friendswood, Dickinson, Kemah, Galveston, Texas City, and surrounding communities.
What Is SPRAVATO®?
SPRAVATO® is a prescription nasal spray medication containing esketamine, a compound related to ketamine. It is the first FDA-approved nasal spray treatment specifically developed for treatment-resistant depression and for major depressive disorder with acute suicidal ideation or behavior.
Unlike traditional antidepressants that primarily affect serotonin or norepinephrine, SPRAVATO® works on the brain's glutamate system by targeting NMDA (N-methyl-D-aspartate) receptors involved in mood regulation and neural connectivity. This unique mechanism allows SPRAVATO® to produce antidepressant effects more rapidly than conventional medications for many patients.
SPRAVATO® was approved by the FDA in 2019 and is manufactured by Janssen Pharmaceuticals, a Johnson & Johnson company. It is available only through a certified healthcare provider as part of the SPRAVATO REMS (Risk Evaluation and Mitigation Strategy) program to ensure patient safety during administration.
What Conditions Does SPRAVATO® Treat?
SPRAVATO® is FDA-approved for adults with:
Treatment-Resistant Depression (TRD) Depression that has not improved despite trying two or more antidepressant medications of adequate dose and duration.
Major Depressive Disorder (MDD) with acute suicidal thoughts or behaviors, For adults with MDD who are experiencing active suicidal thoughts or behaviors, when used in combination with an oral antidepressant.
How Does SPRAVATO® Treatment Work?
SPRAVATO® must be administered only in a certified medical office under close supervision, it cannot be taken home. At Family Psychiatry in League City, TX, our clinical team guides patients through every step of the process in a dedicated, comfortable treatment room.
The treatment schedule:
During the initial induction phase (weeks 1–4), SPRAVATO® is typically administered twice per week. During the maintenance phase (weeks 5–8), sessions are reduced to once per week. After week 8, ongoing maintenance treatment is once every one to two weeks, based on your response and your treatment plan developed by Dr. Saeed.
What happens during each session:
You arrive at Family Psychiatry and are checked in by our clinical team
Your blood pressure is measured before and after treatment
You self-administer the nasal spray under provider supervision
You rest comfortably in our SPRAVATO® treatment room for a minimum of two hours while our team monitors you for side effects including dissociation, dizziness, or sedation
Once cleared by clinical staff, a responsible adult must drive you home — you cannot drive or operate heavy machinery until the following day
SPRAVATO® is always used in combination with an oral antidepressant, not as a standalone treatment.
Is SPRAVATO® Safe?
Yes! SPRAVATO® is considered safe and effective when administered in a certified healthcare setting under proper medical supervision. Because of the potential for temporary dissociation and sedation, it is only available through the SPRAVATO REMS program, which requires all treatment to occur in a supervised clinical environment.
Family Psychiatry in League City, TX is a certified SPRAVATO® treatment center. Dr. Mohammad Saeed evaluates every patient prior to beginning treatment to ensure SPRAVATO® is appropriate and safe for their specific medical history and circumstances.
Possible Side Effects May Include:
Dissociation - feeling detached, dreamlike, or "spaced out" (most common, typically resolves within 2 hours)
Dizziness or vertigo
Nausea
Sedation or sleepiness
Temporary increase in blood pressure (monitored closely during each session)
Headache
These side effects typically occur shortly after dosing and resolve during the 2-hour monitoring period at our clinic. Because of these effects, patients must be accompanied by a responsible adult who can drive them home after each session.
SPRAVATO® may not be appropriate for individuals with:
Uncontrolled hypertension or a history of intracerebral hemorrhage
A history of psychosis or certain other psychiatric conditions
Active substance use disorder
Certain cardiovascular conditions
Aneurysmal vascular disease
A thorough medical and psychiatric evaluation by Dr. Saeed is required before SPRAVATO® treatment begins at Family Psychiatry.
Does SPRAVATO® Work?
Yes! SPRAVATO® has demonstrated significant clinical effectiveness in treating treatment-resistant depression, particularly for patients who have not responded to multiple antidepressant medications.
What the clinical evidence shows:
In Phase 3 clinical trials, SPRAVATO® combined with a new oral antidepressant showed statistically significant improvement in depressive symptoms compared to placebo plus antidepressant
Many patients experience a meaningful reduction in depressive symptoms within the first 24 hours of treatment - significantly faster than traditional antidepressants, which typically take 4–6 weeks
For patients with MDD and acute suicidal ideation, SPRAVATO® demonstrated significant reduction in suicidal thinking within 24 hours
Clinical response rates in real-world settings are consistent with trial data, with many patients achieving sustained remission
One of SPRAVATO®'s most meaningful clinical advantages is its speed of action. For patients in acute distress or with treatment-resistant symptoms, the ability to experience relief within hours rather than weeks can be life-changing.
As with all depression treatments, individual results vary. Ongoing follow-up care and continued oral antidepressant use are essential to maintaining improvement.
Everything You Need To Know About Spravato®
SPRAVATO® (esketamine) nasal spray is a rapid-acting, FDA-approved treatment for adults with treatment-resistant depression (TRD) and for those with major depressive disorder (MDD) with acute suicidal ideation or behavior.
With thousands of patients treated under strict medical supervision, SPRAVATO® is providing new hope for individuals living with depression, helping many achieve meaningful improvement in mood and overall functioning.
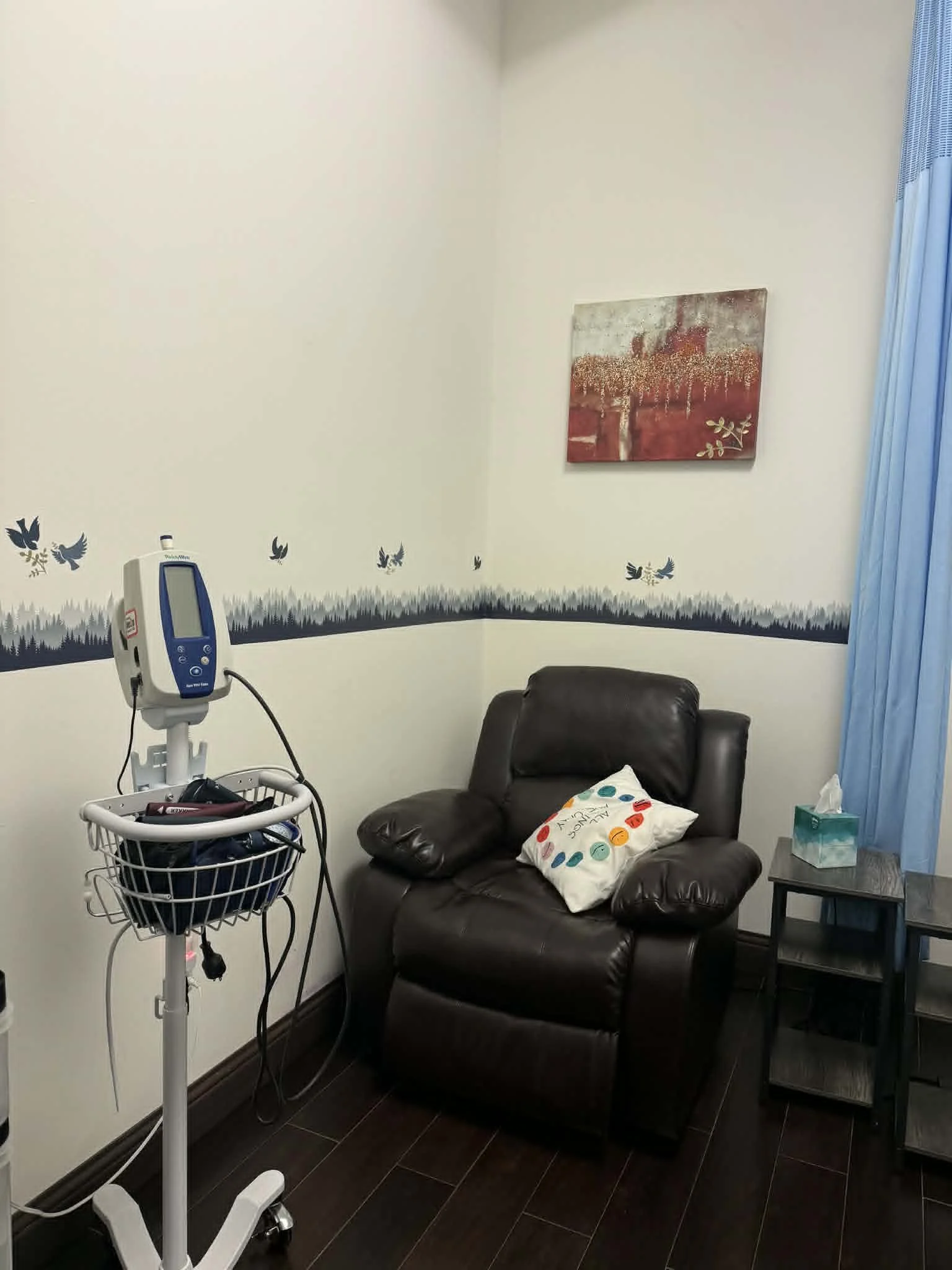
What to Expect in Our SPRAVATO® Treatment Room:
What to Expect at Family Psychiatry's SPRAVATO® Treatment Room
Our dedicated SPRAVATO® treatment room at Family Psychiatry in League City, TX is designed to provide a safe, calm, and comfortable environment throughout your session.
Before your first appointment: Dr. Mohammad Saeed will conduct a complete psychiatric evaluation to confirm your diagnosis, review your antidepressant history, assess your medical background, and determine whether SPRAVATO® is appropriate for you. If approved, our team will walk you through the entire process, answer your questions, and schedule your first session.
On the day of each treatment:
Arrange for a responsible adult to drive you to and from your appointment — you cannot drive on the day of treatment
Do not eat for at least 2 hours before your session (light meals only if needed)
Avoid taking nasal decongestants or nasal steroids on treatment days
Arrive at Family Psychiatry's League City clinic at your scheduled time
During your session:
Our clinical team will measure your blood pressure and check in on how you are feeling
You will self-administer the SPRAVATO® nasal spray under provider guidance
You will rest comfortably in our dedicated treatment room in a reclined chair
Our staff monitors you continuously for the full 2-hour observation period
Most patients experience some degree of dissociation - this is expected and temporary
After your session:
You are cleared to leave once clinical staff confirm it is safe to do so
Your accompanying adult will drive you home
Do not drive, operate machinery, or make major decisions until the following day
Most patients are able to return to normal activities the next morning
SPRAVATO® VS TMS - WHICH IS RIGHT FOR YOU?
Family Psychiatry in League City, TX offers both SPRAVATO® and NeuroStar TMS Therapy for patients with treatment-resistant depression. Both are advanced, FDA-approved options — but they work differently and suit different patients.
SPRAVATO® may be a better fit if you:
Need rapid symptom relief (Spravato can work within hours to days)
Are experiencing active suicidal ideation alongside depression
Are able to commit to twice-weekly in-office sessions during the initial phase
Can arrange transportation on treatment days (driving is not permitted)
TMS therapy may be a better fit if you:
Prefer a fully non-medication approach
Want to avoid systemic side effects entirely
Are able to commit to daily sessions 5 days per week for 7 weeks
Do not have metal implants or conditions that contraindicate TMS
Some patients may be candidates for both treatments depending on their diagnosis and history. Dr. Mohammad Saeed will conduct a thorough evaluation and recommend the most appropriate option — or combination — for your individual situation.
Learn more about TMS Therapy at Family Psychiatry →
Does Insurance Cover SPRAVATO®?
Many insurance plans, including Medicare and most major commercial carriers, may cover SPRAVATO® for eligible patients with treatment-resistant depression. Coverage depends on your specific plan and whether you meet the clinical criteria — typically having tried at least two antidepressants without adequate relief.
Our team at Family Psychiatry in League City, TX will assist with insurance verification and prior authorization before you begin treatment so you have a clear picture of your coverage and out-of-pocket costs.
To verify your coverage: Call our office at 281-554-0123 or visit our insurance page. We are here to make the process as straightforward as possible.
Frequently Asked Questions:
-
SPRAVATO® (esketamine) is an FDA-approved nasal spray treatment for two conditions: treatment-resistant depression (TRD) in adults, defined as depression that has not improved after trying two or more antidepressant medications, and major depressive disorder (MDD) with acute suicidal ideation or behavior. Family Psychiatry in League City, TX offers SPRAVATO® treatment under the supervision of Dr. Mohammad Saeed, M.D. Call 281-554-0123 to find out if you qualify.
-
Traditional antidepressants such as SSRIs and SNRIs work by increasing serotonin or norepinephrine levels in the brain. SPRAVATO® works differently, it targets the glutamate system by blocking NMDA receptors, which plays a key role in mood regulation and neural plasticity. This different mechanism allows SPRAVATO® to act much faster than conventional antidepressants. Many patients notice improvement within hours to days rather than the 4 to 6 weeks typical of oral antidepressants.
-
SPRAVATO® is administered as a nasal spray in a certified medical setting under clinical supervision. At Family Psychiatry in League City, TX, patients self-administer the nasal spray under Dr. Saeed's team's guidance, then remain in our dedicated SPRAVATO® treatment room for a minimum of two hours while staff monitor for side effects including dissociation, dizziness, or changes in blood pressure. Patients must arrange for a responsible adult to drive them home and should not drive until the following day.
-
Yes. Clinical trials show that SPRAVATO® combined with an oral antidepressant produces statistically significant improvement in depressive symptoms in adults with treatment-resistant depression. Many patients experience meaningful symptom relief within the first 24 hours of treatment, much faster than traditional antidepressants. At Family Psychiatry in League City, TX, Dr. Mohammad Saeed evaluates each patient individually and monitors their progress throughout the full treatment course.
-
SPRAVATO® treatment at Family Psychiatry in League City, TX follows a phased schedule. During the induction phase (weeks 1–4), sessions are scheduled twice per week. During the optimization phase (weeks 5–8), sessions are reduced to once per week. For ongoing maintenance (week 9 and beyond), sessions are typically every one to two weeks depending on your response and Dr. Saeed's clinical recommendation.
-
The most common side effects of SPRAVATO® include dissociation (a feeling of being detached or dreamlike), dizziness, nausea, sedation, and a temporary increase in blood pressure. These effects typically occur shortly after administration and resolve during the 2-hour monitoring period at Family Psychiatry in League City, TX. Because of these effects, patients cannot drive on the day of treatment and must be accompanied by a responsible adult.
-
Many insurance plans including Medicare and major commercial carriers may cover SPRAVATO® for eligible patients with treatment-resistant depression. Coverage depends on your specific plan and medical history. Family Psychiatry in League City, TX assists patients with insurance verification and prior authorization before starting treatment. Contact our office at 281-554-0123 or visit family-psychiatry.co/insurances-accepted to check your coverage.
-
SPRAVATO® is one of the fastest-acting treatments available for depression. Many patients experience a meaningful reduction in depressive symptoms within hours to days of their first treatment session. This is significantly faster than traditional antidepressants, which typically take 4 to 6 weeks to show full effect. For patients with MDD and acute suicidal ideation, clinical trials demonstrated significant reduction in suicidal thinking within 24 hours of treatment.
-
Family Psychiatry offers SPRAVATO® treatment at our certified clinic located at 2360 Gulf Freeway S, Suite 100B, League City, TX 77573. All treatments are evaluated and supervised by Dr. Mohammad Saeed, M.D., a triple board-certified psychiatrist. We serve patients from League City, Clear Lake, Friendswood, Dickinson, Kemah, Galveston, Texas City, and the Greater Houston area. Call 281-554-0123 or visit family-psychiatry.co/appointment-request to schedule a consultation.
-
This depends on your individual clinical situation. Both SPRAVATO® and TMS therapy are advanced treatments for depression, and in some cases a provider may recommend one over the other or discuss a sequenced approach. At Family Psychiatry in League City, TX, Dr. Mohammad Saeed will evaluate your full history and make a personalized recommendation. Learn more about TMS therapy at family-psychiatry.co/tms.
Ready to Find Out If Spravato® Is Right for You?
If you or a loved one are living with treatment-resistant depression and have not found relief from antidepressant medications, SPRAVATO® at Family Psychiatry in League City, TX may offer the relief you have been looking for. Dr. Mohammad Saeed and our clinical team are here to evaluate your situation, answer your questions, and guide you through every step of the treatment process.
Call us today: 281-554-0123 Request a consultation: Online Appointment Form
Important Safety Information:
SPRAVATO® is available by prescription only and must be administered in a certified healthcare setting. Patients are monitored after each treatment due to risks of sedation, dissociation, and increased blood pressure. SPRAVATO® is not appropriate for everyone.
Your provider will determine whether this treatment is suitable based on your medical history and current condition.
SPRAVATO® Is Indicated For:
Treatment-Resistant Depression (TRD) in adults
Depressive symptoms in adults with Major Depressive Disorder (MDD) who experience acute suicidal ideation or behavior
SPRAVATO® must be administered in a certified healthcare setting under medical supervision as part of a federally required safety program.
For additional prescribing and safety information, visit:
👉 www.SPRAVATOrems.com